
An assessment of histone-modification antibody quality. Immunofluorescence and fluorescent-protein tagging show high correlation for protein localization in mammalian cells. Drug development: raise standards for preclinical cancer research. 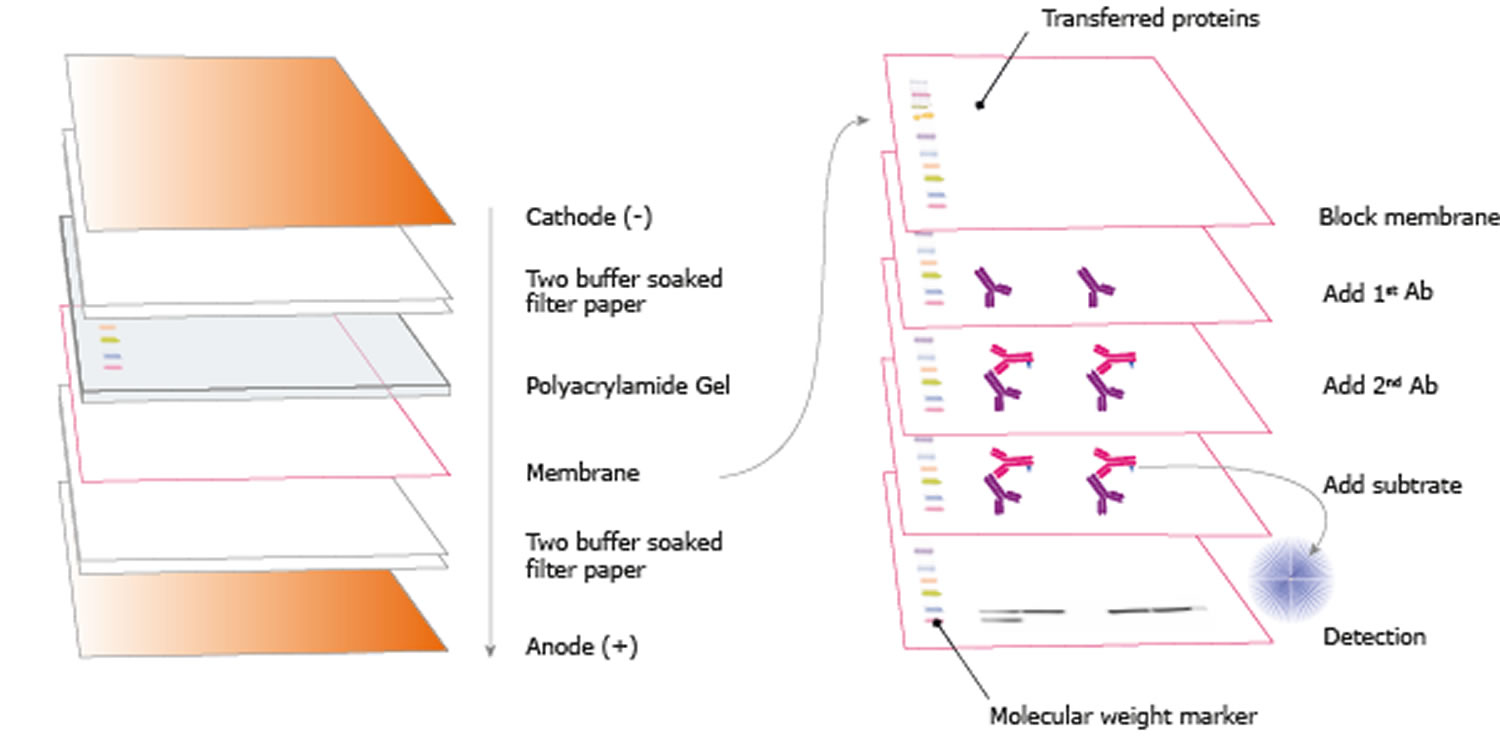
Single-cell western blotting after whole-cell imaging to assess cancer chemotherapeutic response. Tracking lineages of single cells in lines using a microfluidic device. Rowat, A.C., Bird, J.C., Agresti, J.J., Rando, O.J. Causal protein-signaling networks derived from multiparameter single-cell data. Sachs, K., Perez, O., Pe'er, D., Lauffenburger, D.A. coli proteome and transcriptome with single-molecule sensitivity in single cells. Sequence signatures and mRNA concentration can explain two-thirds of protein abundance variation in a human cell line. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. RNA sequencing of pancreatic circulating tumour cells implicates WNT signalling in metastasis. Microfluidic single-cell whole-transcriptome sequencing. High-throughput microfluidic single-cell RT-qPCR. Combinatorial labeling of single cells for gene expression cytometry. Scalable microfluidics for single cell RNA printing and sequencing. Microfluidic single-cell mRNA isolation and analysis. Microfluidics: reframing biological enquiry. Integrated microfluidic bioprocessor for single-cell gene expression analysis. Automated single-cell motility analysis on a chip using lensfree microscopy. Dynamic proteomics of individual cancer cells in response to a drug. Transcriptome-wide noise controls lineage choice in mammalian progenitor cells. Genome-wide single-cell analysis of recombination activity and de novo mutation rates in human sperm. The technique is relevant when direct measurement of proteins in single cells is needed, with applications spanning the fundamental biosciences to applied biomedicine. Once the microdevice has been fabricated, the assay can be completed in 4–6 h by microfluidic novices and it generates high-selectivity, multiplexed data from single cells. To extend in-gel immunoprobing to gels of small pore size, we describe an optional gel de-cross-linking protocol for more effective introduction of antibodies into the gel layer. We also describe microdevice fabrication for both uniform and pore-gradient microgels. Multiplexing can be achieved by probing with antibody cocktails and using antibody stripping/reprobing techniques, enabling detection of 10+ proteins in each cell. scWB involves five main stages: (i) gravity settling of cells into microwells (ii) chemical lysis of cells in each microwell (iii) PAGE of each single-cell lysate (iv) exposure of the gel to UV light to blot (immobilize) proteins to the gel matrix and (v) in-gel immunoprobing of immobilized proteins. The gel layer functions as both a molecular sieving matrix during PAGE and a blotting scaffold during immunoprobing. scWB is performed on a microdevice that comprises an array of microwells molded in a thin layer of a polyacrylamide gel (PAG). Like conventional western blotting, single-cell western blotting (scWB) is particularly useful for protein targets that lack selective antibodies (e.g., isoforms) and in cases in which background signal from intact cells is confounding. This protocol describes how to perform western blotting on individual cells to measure cell-to-cell variation in protein expression levels and protein state.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
